Metaglip Treatment for Diabetes - Metaglip Full Prescribing Information
Brand Name: Metaglip
Generic Name: Glipizide and Metformin Hydrochloride
Dosage Form: tablet, film coated
Metaglip™ (glipizide and metformin HCl) Tablets
- 2.5 mg/250 mg
- 2.5 mg/500 mg
- 5 mg/500 mg
Contents:
Description
Clinical Pharmacology
Indications and Usage
Contraindications
Warnings
Precautions
Adverse Reactions
Overdose
Dosage and Administration
How Supplied
Metaglip, glipizide and metformin hydrochloride, patient information sheet (in plain English)
Description
Metaglip™ (glipizide and metformin HCl) Tablets contain 2 oral antihyperglycemic drugs used in the management of type 2 diabetes, glipizide and metformin hydrochloride.
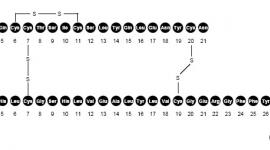
Glipizide is an oral antihyperglycemic drug of the sulfonylurea class. The chemical name for glipizide is 1-cyclohexyl-3-[[p-[2-(5-methylpyrazinecarboxamido)ethyl]phenyl]sulfonyl]urea. Glipizide is a whitish, odorless powder with a molecular formula of C21H27N5O4S, a molecular weight of 445.55 and a pKa of 5.9. It is insoluble in water and alcohols, but soluble in 0.1 N NaOH; it is freely soluble in dimethylformamide. The structural formula is represented below.

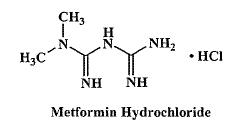
Metformin hydrochloride is an oral antihyperglycemic drug used in the management of type 2 diabetes. Metformin hydrochloride (N,N-dimethylimidodicarbonimidic diamide monohydrochloride) is not chemically or pharmacologically related to sulfonylureas, thiazolidinediones, or α-glucosidase inhibitors. It is a white to off-white crystalline compound with a molecular formula of C4H12ClN5 (monohydrochloride) and a molecular weight of 165.63. Metformin hydrochloride is freely soluble in water and is practically insoluble in acetone, ether, and chloroform. The pKa of metformin is 12.4. The pH of a 1% aqueous solution of metformin hydrochloride is 6.68. The structural formula is as shown:
Metaglip is available for oral administration in tablets containing 2.5 mg glipizide with 250 mg metformin hydrochloride, 2.5 mg glipizide with 500 mg metformin hydrochloride, and 5 mg glipizide with 500 mg metformin hydrochloride. In addition, each tablet contains the following inactive ingredients: microcrystalline cellulose, povidone, croscarmellose sodium, and magnesium stearate. The tablets are film coated, which provides color differentiation.
Clinical Pharmacology
Mechanism of Action
Metaglip combines glipizide and metformin hydrochloride, 2 antihyperglycemic agents with complementary mechanisms of action, to improve glycemic control in patients with type 2 diabetes.
Glipizide appears to lower blood glucose acutely by stimulating the release of insulin from the pancreas, an effect dependent upon functioning beta cells in the pancreatic islets. Extrapancreatic effects may play a part in the mechanism of action of oral sulfonylurea hypoglycemic drugs. The mechanism by which glipizide lowers blood glucose during long-term administration has not been clearly established. In man, stimulation of insulin secretion by glipizide in response to a meal is undoubtedly of major importance. Fasting insulin levels are not elevated even on long-term glipizide administration, but the postprandial insulin response continues to be enhanced after at least 6 months of treatment.
Metformin hydrochloride is an antihyperglycemic agent that improves glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Metformin hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization.
Pharmacokinetics
Absorption and Bioavailability
Metaglip
In a single-dose study in healthy subjects, the glipizide and metformin components of Metaglip 5 mg/500 mg were bioequivalent to coadministered GLUCOTROL® and GLUCOPHAGE®. Following administration of a single Metaglip 5 mg/500 mg tablet in healthy subjects with either a 20% glucose solution or a 20% glucose solution with food, there was a small effect of food on peak plasma concentration (Cmax) and no effect of food on area under the curve (AUC) of the glipizide component. Time to peak plasma concentration (Tmax) for the glipizide component was delayed 1 hour with food relative to the same tablet strength administered fasting with a 20% glucose solution. Cmax for the metformin component was reduced approximately 14% by food whereas AUC was not affected. Tmax for the metformin component was delayed 1 hour after food.
Glipizide
Gastrointestinal absorption of glipizide is uniform, rapid, and essentially complete. Peak plasma concentrations occur 1 to 3 hours after a single oral dose. Glipizide does not accumulate in plasma on repeated oral administration. Total absorption and disposition of an oral dose was unaffected by food in normal volunteers, but absorption was delayed by about 40 minutes.
Metformin Hydrochloride
The absolute bioavailability of a 500 mg metformin hydrochloride tablet given under fasting conditions is approximately 50% to 60%. Studies using single oral doses of metformin tablets of 500 mg and 1500 mg, and 850 mg to 2550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination. Food decreases the extent of and slightly delays the absorption of metformin, as shown by approximately a 40% lower peak concentration and a 25% lower AUC in plasma and a 35-minute prolongation of time to peak plasma concentration following administration of a single 850 mg tablet of metformin with food, compared to the same tablet strength administered fasting. The clinical relevance of these decreases is unknown.
Distribution
Glipizide
Protein binding was studied in serum from volunteers who received either oral or intravenous glipizide and found to be 98% to 99% 1 hour after either route of administration. The apparent volume of distribution of glipizide after intravenous administration was 11 liters, indicative of localization within the extracellular fluid compartment. In mice, no glipizide or metabolites were detectable autoradiographically in the brain or spinal cord of males or females, nor in the fetuses of pregnant females. In another study, however, very small amounts of radioactivity were detected in the fetuses of rats given labeled drug.
Metformin Hydrochloride
The apparent volume of distribution (V/F) of metformin following single oral doses of 850 mg averaged 654 ±358 L. Metformin is negligibly bound to plasma proteins. Metformin partitions into erythrocytes, most likely as a function of time. At usual clinical doses and dosing schedules of metformin, steady state plasma concentrations of metformin are reached within 24 to 48 hours and are generally
Metabolism and Elimination
Glipizide
The metabolism of glipizide is extensive and occurs mainly in the liver. The primary metabolites are inactive hydroxylation products and polar conjugates, and are excreted mainly in the urine. Less than 10% unchanged glipizide is found in the urine. The half-life of elimination ranges from 2 to 4 hours in normal subjects, whether given intravenously or orally. The metabolic and excretory patterns are similar with the 2 routes of administration, indicating that first-pass metabolism is not significant.
Metformin Hydrochloride
Intravenous single-dose studies in normal subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) nor biliary excretion. Renal clearance (see Table 1) is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Special Populations
Patients With Type 2 Diabetes
In the presence of normal renal function, there are no differences between single- or multiple-dose pharmacokinetics of metformin between patients with type 2 diabetes and normal subjects (see Table 1), nor is there any accumulation of metformin in either group at usual clinical doses.
Hepatic Insufficiency
The metabolism and excretion of glipizide may be slowed in patients with impaired hepatic function (see PRECAUTIONS). No pharmacokinetic studies have been conducted in patients with hepatic insufficiency for metformin.
Renal Insufficiency
The metabolism and excretion of glipizide may be slowed in patients with impaired renal function (see PRECAUTIONS).
In patients with decreased renal function (based on creatinine clearance), the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased in proportion to the decrease in creatinine clearance (see Table 1; also, see WARNINGS).
Geriatrics
There is no information on the pharmacokinetics of glipizide in elderly patients.
Limited data from controlled pharmacokinetic studies of metformin in healthy elderly subjects suggest that total plasma clearance is decreased, the half-life is prolonged, and Cmax is increased, when compared to healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function (see Table 1). Metformin treatment should not be initiated in patients ≥80 years of age unless measurement of creatinine clearance demonstrates that renal function is not reduced.
Table 1: Select Mean ( ±SD) Metformin Pharmacokinetic Parameters Following Single or Multiple Oral Doses of Metformin
| Subject Groups: Metformin Dosea (Number of Subjects) | Cmaxb ( µg/mL) | Tmaxc (hrs) | Renal Clearance (mL/min) |
|---|---|---|---|
| Healthy, Nondiabetic Adults: | |||
| 500 mg SDd (24) | 1.03 ( ±0.33) | 2.75 ( ±0.81) | 600 ( ±132) |
| 850 mg SD (74)e | 1.60 ( ±0.38) | 2.64 ( ±0.82) | 552 ( ±139) |
| 850 mg t.i.d. for 19 dosesf (9) | 2.01 ( ±0.42) | 1.79 ( ±0.94) | 642 ( ±173) |
| Adults with Type 2 Diabetes: | |||
| 850 mg SD (23) | 1.48 ( ±0.5) | 3.32 ( ±1.08) | 491 ( ±138) |
| 850 mg t.i.d. for 19 dosesf (9) | 1.90 ( ±0.62) | 2.01 ( ±1.22) | 550 ( ±160) |
| Elderlyg, Healthy Nondiabetic Adults: | |||
| 850 mg SD (12) | 2.45 ( ±0.70) | 2.71 ( ±1.05) | 412 ( ±98) |
| Renal-impaired Adults: 850 mg SD | |||
| Mild (CLcrh 61-90 mL/min) (5) | 1.86 ( ±0.52) | 3.20 ( ±0.45) | 384 ( ±122) |
| Moderate (CLcr 31-60 mL/min) (4) | 4.12 ( ±1.83) | 3.75 ( ±0.50) | 108 ( ±57) |
| Severe (CLcr 10-30 mL/min) (6) | 3.93 ( ±0.92) | 4.01 ( ±1.10) | 130 ( ±90) |
| a All doses given fasting except the first 18 doses of the multiple-dose studies | |||
| b Peak plasma concentration | |||
| c Time to peak plasma concentration | |||
| d SD=single dose | |||
| e Combined results (average means) of 5 studies: mean age 32 years (range 23-59 years) | |||
| f Kinetic study done following dose 19, given fasting | |||
| g Elderly subjects, mean age 71 years (range 65-81 years) | |||
| h CLcr=creatinine clearance normalized to body surface area of 1.73 m2 | |||
Pediatrics
No data from pharmacokinetic studies in pediatric subjects are available for glipizide.
After administration of a single oral GLUCOPHAGE 500 mg tablet with food, geometric mean metformin Cmax and AUC differed <5% between pediatric type 2 diabetic patients (12-16 years of age) and gender- and weight-matched healthy adults (20-45 years of age), all with normal renal function.
Gender
There is no information on the effect of gender on the pharmacokinetics of glipizide.
Metformin pharmacokinetic parameters did not differ significantly in subjects with or without type 2 diabetes when analyzed according to gender (males=19, females=16). Similarly, in controlled clinical studies in patients with type 2 diabetes, the antihyperglycemic effect of metformin was comparable in males and females.
Race
No information is available on race differences in the pharmacokinetics of glipizide.
No studies of metformin pharmacokinetic parameters according to race have been performed. In controlled clinical studies of metformin in patients with type 2 diabetes, the antihyperglycemic effect was comparable in whites (n=249), blacks (n=51), and Hispanics (n=24).
Clinical Studies
Patients with Inadequate Glycemic Control on Diet and Exercise Alone
In a 24-week, double-blind, active-controlled, multicenter international clinical trial, patients with type 2 diabetes, whose hyperglycemia was not adequately controlled with diet and exercise alone (hemoglobin A1c [HbA1c] >7.5% and ≤12%, and fasting plasma glucose [FPG]
Table 2: Active-Controlled Trial of Metaglip in Patients with Inadequate Glycemic Control on Diet and Exercise Alone: Summary of Trial Data at 24 Weeks
| Glipizide 5 mg tablets | Metformin 500 mg tablets | Metaglip 2.5 mg/250 mg tablets | Metaglip 2.5 mg/500 mg tablets | |
|---|---|---|---|---|
| ap<0.001 | ||||
| Mean Final Dose | 16.7 mg | 1749 mg | 7.9 mg/ 791 mg | 7.4 mg/ 1477 mg |
| Hemoglobin A1c (%) | N=168 | N=171 | N=166 | N=163 |
| Baseline Mean | 9.17 | 9.15 | 9.06 | 9.10 |
| Final Mean | 7.36 | 7.67 | 6.93 | 6.95 |
| Adjusted Mean Change from Baseline | −1.77 | −1.46 | −2.15 | −2.14 |
| Difference from Glipizide | −0.38a | −0.37a | ||
| Difference from Metformin | −0.70a | −0.69a | ||
| % Patients with Final HbA1c <7% | 43.5% | 35.1% | 59.6% | 57.1% |
| Fasting Plasma Glucose (mg/dL) | N=169 | N=176 | N=170 | N=169 |
| Baseline Mean | 210.7 | 207.4 | 206.8 | 203.1 |
| Final Mean | 162.1 | 163.8 | 152.1 | 148.7 |
| Adjusted Mean Change from Baseline | −46.2 | −42.9 | −54.2 | −56.5 |
| Difference from Glipizide | −8.0 | −10.4 | ||
| Difference from Metformin | −11.3 | −13.6 | ||
After 24 weeks, treatment with Metaglip 2.5 mg/250 mg and 2.5 mg/500 mg resulted in significantly greater reduction in HbA1c compared to glipizide and metformin therapy. Also, Metaglip 2.5 mg/250 mg therapy resulted in significant reductions in FPG versus metformin therapy.
Increases above fasting glucose and insulin levels were determined at baseline and final study visits by measurement of plasma glucose and insulin for 3 hours following a standard mixed liquid meal. Treatment with Metaglip lowered the 3-hour postprandial glucose AUC, compared to baseline, to a significantly greater extent than did the glipizide and the metformin therapies. Compared to baseline, Metaglip enhanced the postprandial insulin response, but did not significantly affect fasting insulin levels.
There were no clinically meaningful differences in changes from baseline for all lipid parameters between Metaglip therapy and either metformin therapy or glipizide therapy. The adjusted mean changes from baseline in body weight were: Metaglip 2.5 mg/250 mg, −0.4 kg; Metaglip 2.5 mg/500 mg, −0.5 kg; glipizide, −0.2 kg; and metformin, −1.9 kg. Weight loss was greater with metformin than with Metaglip.
Patients with Inadequate Glycemic Control on Sulfonylurea Monotherapy
In an 18-week, double-blind, active-controlled U.S. clinical trial, a total of 247 patients with type 2 diabetes not adequately controlled (HbA1c ≥7.5% and ≤12%, and FPG
In an 18-week, double-blind, active-controlled U.S. clinical trial, a total of 247 patients with type 2 diabetes not adequately controlled (HbA1c ≥7.5% and ≤12%, and FPG
Table 3: Metaglip in Patients with Inadequate Glycemic Control on Sulfonylurea Alone: Summary of Trial Data at 18 Weeks
| Glipizide 5 mg tablets | Metformin 500 mg tablets | Metaglip 5 mg/500mg tablets | |
|---|---|---|---|
| ap<0.001 | |||
| Mean Final Dose | 30.0 mg | 1927 mg | 17.5 mg/ 1747 mg |
| Hemoglobin A1c(%) | N=79 | N=71 | N=80 |
| Baseline Mean | 8.87 | 8.61 | 8.66 |
| Final Adjusted Mean | 8.45 | 8.36 | 7.39 |
| Difference from Glipizide | −1.06a | ||
| Difference from Metformin | −0.98a | ||
| % Patients with Final HbA1c <7% | 8.9% | 9.9% | 36.3% |
| Fasting Plasma Glucose (mg/dL) | N=82 | N=75 | N=81 |
| Baseline Mean | 203.6 | 191.3 | 194.3 |
| Adjusted Mean Change from Baseline | 7.0 | 6.7 | −30.4 |
| Difference from Glipizide | −37.4 | ||
| Difference from Metformin | −37.2 | ||
After 18 weeks, treatment with Metaglip at doses up to 20 mg/2000 mg per day resulted in significantly lower mean final HbA1c and significantly greater mean reductions in FPG compared to glipizide and metformin therapy. Treatment with Metaglip lowered the 3-hour postprandial glucose AUC, compared to baseline, to a significantly greater extent than did the glipizide and the metformin therapies. Metaglip did not significantly affect fasting insulin levels.
There were no clinically meaningful differences in changes from baseline for all lipid parameters between Metaglip therapy and either metformin therapy or glipizide therapy. The adjusted mean changes from baseline in body weight were: Metaglip 5 mg/500 mg, −0.3 kg; glipizide, −0.4 kg; and metformin, −2.7 kg. Weight loss was greater with metformin than with Metaglip.
Indications and Usage
Metaglip (glipizide and metformin HCl) Tablets is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Contraindications
Metaglip is contraindicated in patients with:
- Renal disease or renal dysfunction (eg, as suggested by serum creatinine levels ≥1.5 mg/dL [males], ≥1.4 mg/dL [females], or abnormal creatinine clearance) which may also result from conditions such as cardiovascular collapse (shock), acute myocardial infarction, and septicemia (see WARNINGS and PRECAUTIONS).
- Known hypersensitivity to glipizide or metformin hydrochloride.
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis, with or without coma. Diabetic ketoacidosis should be treated with insulin.
Metaglip should be temporarily discontinued in patients undergoing radiologic studies involving intravascular administration of iodinated contrast materials, because use of such products may result in acute alteration of renal function. (See also PRECAUTIONS.)
Warnings
Metformin Hydrochloride
Lactic acidosis
Lactic acidosis is a rare, but serious, metabolic complication that can occur due to metformin accumulation during treatment with METAGLIP; when it occurs, it is fatal in approximately 50% of cases. Lactic acidosis may also occur in association with a number of pathophysiologic conditions, including diabetes mellitus, and whenever there is significant tissue hypoperfusion and hypoxemia. Lactic acidosis is characterized by elevated blood lactate levels ( > 5 mmol/L), decreased blood pH, electrolyte disturbances with an increased anion gap, and an increased lactate/pyruvate ratio. When metformin is implicated as the cause of lactic acidosis, metformin plasma levels > 5 μg/mL are generally found.
The reported incidence of lactic acidosis in patients receiving metformin hydrochloride is very low (approximately 0.03 cases/1000 patient-years, with approximately 0.015 fatal cases/1000 patient-years). In more than 20,000 patient-years exposure to metformin in clinical trials, there were no reports of lactic acidosis. Reported cases have occurred primarily in diabetic patients with significant renal insufficiency, including both intrinsic renal disease and renal hypoperfusion, often in the setting of multiple concomitant medical/surgical problems and multiple concomitant medications. Patients with congestive heart failure requiring pharmacologic management, in particular those with unstable or acute congestive heart failure who are at risk of hypoperfusion and hypoxemia, are at increased risk of lactic acidosis. The risk of lactic acidosis increases with the degree of renal dysfunction and the patient's age. The risk of lactic acidosis may, therefore, be significantly decreased by regular monitoring of renal function in patients taking metformin and by use of the minimum effective dose of metformin. In particular, treatment of the elderly should be accompanied by careful monitoring of renal function. METAGLIP treatment should not be initiated in patients ≥ 80 years of age unless measurement of creatinine clearance demonstrates that renal function is not reduced, as these patients are more susceptible to developing lactic acidosis. In addition, METAGLIP should be promptly withheld in the presence of any condition associated with hypoxemia, dehydration, or sepsis. Because impaired hepatic function may significantly limit the ability to clear lactate, METAGLIP should generally be avoided in patients with clinical or laboratory evidence of hepatic disease. Patients should be cautioned against excessive alcohol intake, either acute or chronic, when taking METAGLIP, since alcohol potentiates the effects of metformin hydrochloride on lactate metabolism. In addition, METAGLIP should be temporarily discontinued prior to any intravascular radiocontrast study and for any surgical procedure (see also PRECAUTIONS).
The onset of lactic acidosis often is subtle, and accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, increasing somnolence, and nonspecific abdominal distress. There may be associated hypothermia, hypotension, and resistant bradyarrhythmias with more marked acidosis. The patient and the patient's physician must be aware of the possible importance of such symptoms and the patient should be instructed to notify the physician immediately if they occur (see also PRECAUTIONS). METAGLIP should be withdrawn until the situation is clarified. Serum electrolytes, ketones, blood glucose, and if indicated, blood pH, lactate levels, and even blood metformin levels may be useful. Once a patient is stabilized on any dose level of METAGLIP, gastrointestinal symptoms, which are common during initiation of therapy with metformin, are unlikely to be drug related. Later occurrence of gastrointestinal symptoms could be due to lactic acidosis or other serious disease.
Levels of fasting venous plasma lactate above the upper limit of normal but less than mmol/L in patients taking METAGLIP do not necessarily indicate impending lactic acidosis and may be explainable by other mechanisms, such as poorly controlled diabetes or obesity, vigorous physical activity, or technical problems in sample handling. (See also PRECAUTIONS.)
Lactic acidosis should be suspected in any diabetic patient with metabolic acidosis lacking evidence of ketoacidosis (ketonuria and ketonemia).
Lactic acidosis is a medical emergency that must be treated in a hospital setting. In a patient with lactic acidosis who is taking METAGLIP, the drug should be discontinued immediately and general supportive measures promptly instituted. Because metformin hydrochloride is dialyzable (with a clearance of up to 170 mL/min under good hemodynamic conditions), prompt hemodialysis is recommended to correct the acidosis and remove the accumulated metformin. Such management often results in prompt reversal of symptoms and recovery. (See also CONTRAINDICATIONS and PRECAUTIONS.)
Special Warning on Increased risk of Cardiovascular Mortality
The administration of oral hypoglycemic drugs has been reported to be associated with increased cardiovascular mortality as compared to treatment with diet alone or diet plus insulin. This warning is based on the study conducted by the University Group Diabetes Program (UGDP), a long-term prospective clinical trial designed to evaluate the effectiveness of glucose-lowering drugs in preventing or delaying vascular complications in patients with non-insulin-dependent diabetes. The study involved 823 patients who were randomly assigned to 1 of 4 treatment groups (Diabetes 19 (Suppl. 2):747-830, 1970).
UGDP reported that patients treated for 5 to 8 years with diet plus a fixed dose of tolbutamide (1.5 grams per day) had a rate of cardiovascular mortality approximately 2 ½ times that of patients treated with diet alone. A significant increase in total mortality was not observed, but the use of tolbutamide was discontinued based on the increase in cardiovascular mortality, thus limiting the opportunity for the study to show an increase in overall mortality. Despite controversy regarding the interpretation of these results, the findings of the UGDP study provide an adequate basis for this warning. The patient should be informed of the potential risks and benefits of glipizide and of alternative modes of therapy.
Although only 1 drug in the sulfonylurea class (tolbutamide) was included in this study, it is prudent from a safety standpoint to consider that this warning may also apply to other hypoglycemic drugs in this class, in view of their close similarities in mode of action and chemical structure.
Precautions
General
Macrovascular Outcomes
There have been no clinical studies establishing conclusive evidence of macrovascular risk reduction with Metaglip or any other antidiabetic drug.
Metaglip
Hypoglycemia
Metaglip is capable of producing hypoglycemia; therefore, proper patient selection, dosing, and instructions are important to avoid potential hypoglycemic episodes. The risk of hypoglycemia is increased when caloric intake is deficient, when strenuous exercise is not compensated by caloric supplementation, or during concomitant use with other glucose-lowering agents or ethanol. Renal insufficiency may cause elevated drug levels of both glipizide and metformin hydrochloride. Hepatic insufficiency may increase drug levels of glipizide and may also diminish gluconeogenic capacity, both of which increase the risk of hypoglycemic reactions. Elderly, debilitated, or malnourished patients and those with adrenal or pituitary insufficiency or alcohol intoxication are particularly susceptible to hypoglycemic effects. Hypoglycemia may be difficult to recognize in the elderly and people who are taking beta-adrenergic blocking drugs.
Glipizide
Renal and hepatic disease
The metabolism and excretion of glipizide may be slowed in patients with impaired renal and/or hepatic function. If hypoglycemia should occur in such patients, it may be prolonged and appropriate management should be instituted.
Hemolytic anemia
Treatment of patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency with sulfonylurea agents can lead to hemolytic anemia. Because Metaglip belongs to the class of sulfonylurea agents, caution should be used in patients with G6PD deficiency and a non-sulfonylurea alternative should be considered. In postmarketing reports, hemolytic anemia has also been reported in patients who did not have known G6PD deficiency.
Metformin Hydrochloride
Monitoring of renal function
Metformin is known to be substantially excreted by the kidney, and the risk of metformin accumulation and lactic acidosis increases with the degree of impairment of renal function. Thus, patients with serum creatinine levels above the upper limit of normal for their age should not receive Metaglip. In patients with advanced age, Metaglip should be carefully titrated to establish the minimum dose for adequate glycemic effect, because aging is associated with reduced renal function. In elderly patients, particularly those ≥80 years of age, renal function should be monitored regularly and, generally, Metaglip should not be titrated to the maximum dose (see WARNINGS and DOSAGE AND ADMINISTRATION). Before initiation of Metaglip therapy and at least annually thereafter, renal function should be assessed and verified as normal. In patients in whom development of renal dysfunction is anticipated, renal function should be assessed more frequently and Metaglip discontinued if evidence of renal impairment is present.
Use of concomitant medications that may affect renal function or metformin disposition
Concomitant medication(s) that may affect renal function or result in significant hemodynamic change or may interfere with the disposition of metformin, such as cationic drugs that are eliminated by renal tubular secretion (see PRECAUTIONS: Drug Interactions), should be used with caution.
Radiologic studies involving the use of intravascular iodinated contrast materials (for example, intravenous urogram, intravenous cholangiography, angiography, and computed tomography (CT) scans with intravascular contrast materials)
Intravascular contrast studies with iodinated materials can lead to acute alteration of renal function and have been associated with lactic acidosis in patients receiving metformin (see CONTRAINDICATIONS). Therefore, in patients in whom any such study is planned, Metaglip should be temporarily discontinued at the time of or prior to the procedure, and withheld for 48 hours subsequent to the procedure and reinstituted only after renal function has been reevaluated and found to be normal.
Hypoxic states
Cardiovascular collapse (shock) from whatever cause, acute congestive heart failure, acute myocardial infarction, and other conditions characterized by hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur in patients on Metaglip therapy, the drug should be promptly discontinued.
Surgical procedures
Metaglip therapy should be temporarily suspended for any surgical procedure (except minor procedures not associated with restricted intake of food and fluids) and should not be restarted until the patient's oral intake has resumed and renal function has been evaluated as normal.
Alcohol intake
Alcohol is known to potentiate the effect of metformin on lactate metabolism. Patients, therefore, should be warned against excessive alcohol intake, acute or chronic, while receiving Metaglip. Due to its effect on the gluconeogenic capacity of the liver, alcohol may also increase the risk of hypoglycemia.
Impaired hepatic function
Since impaired hepatic function has been associated with some cases of lactic acidosis, Metaglip should generally be avoided in patients with clinical or laboratory evidence of hepatic disease.
Vitamin B12 levels
In controlled clinical trials with metformin of 29 weeks duration, a decrease to subnormal levels of previously normal serum vitamin B12, without clinical manifestations, was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex is, however, very rarely associated with anemia and appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Measurement of hematologic parameters on an annual basis is advised in patients on metformin and any apparent abnormalities should be appropriately investigated and managed (see PRECAUTIONS: Laboratory Tests).
Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. In these patients, routine serum vitamin B12 measurements at 2- to 3-year intervals may be useful.
Change in clinical status of patients with previously controlled type 2 diabetes
A patient with type 2 diabetes previously well controlled on metformin who develops laboratory abnormalities or clinical illness (especially vague and poorly defined illness) should be evaluated promptly for evidence of ketoacidosis or lactic acidosis. Evaluation should include serum electrolytes and ketones, blood glucose and, if indicated, blood pH, lactate, pyruvate, and metformin levels. If acidosis of either form occurs, Metaglip must be stopped immediately and other appropriate corrective measures initiated (see also WARNINGS).
Information for Patients
Metaglip
Patients should be informed of the potential risks and benefits of Metaglip and alternative modes of therapy. They should also be informed about the importance of adherence to dietary instructions; a regular exercise program; and regular testing of blood glucose, glycosylated hemoglobin, renal function, and hematologic parameters.
The risks of lactic acidosis associated with metformin therapy, its symptoms, and conditions that predispose to its development, as noted in the WARNINGS and PRECAUTIONS sections, should be explained to patients. Patients should be advised to discontinue Metaglip immediately and promptly notify their health practitioner if unexplained hyperventilation, myalgia, malaise, unusual somnolence, or other nonspecific symptoms occur. Once a patient is stabilized on any dose level of Metaglip, gastrointestinal symptoms, which are common during initiation of metformin therapy, are unlikely to be drug related. Later occurrence of gastrointestinal symptoms could be due to lactic acidosis or other serious disease.
The risks of hypoglycemia, its symptoms and treatment, and conditions that predispose to its development should be explained to patients and responsible family members.
Patients should be counseled against excessive alcohol intake, either acute or chronic, while receiving Metaglip.
Laboratory Tests
Periodic fasting blood glucose (FBG) and HbA1c measurements should be performed to monitor therapeutic response.
Initial and periodic monitoring of hematologic parameters (eg, hemoglobin/hematocrit and red blood cell indices) and renal function (serum creatinine) should be performed, at least on an annual basis. While megaloblastic anemia has rarely been seen with metformin therapy, if this is suspected, vitamin B12 deficiency should be excluded.
Drug Interactions
Metaglip
Certain drugs tend to produce hyperglycemia and may lead to loss of blood glucose control. These drugs include thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid. When such drugs are administered to a patient receiving Metaglip, the patient should be closely observed for loss of blood glucose control. When such drugs are withdrawn from a patient receiving Metaglip, the patient should be observed closely for hypoglycemia. Metformin is negligibly bound to plasma proteins and is, therefore, less likely to interact with highly protein-bound drugs such as salicylates, sulfonamides, chloramphenicol, and probenecid as compared to sulfonylureas, which are extensively bound to serum proteins.
Glipizide
The hypoglycemic action of sulfonylureas may be potentiated by certain drugs, including nonsteroidal anti-inflammatory agents, some azoles, and other drugs that are highly protein-bound, salicylates, sulfonamides, chloramphenicol, probenecid, coumarins, monoamine oxidase inhibitors, and beta-adrenergic blocking agents. When such drugs are administered to a patient receiving Metaglip, the patient should be observed closely for hypoglycemia. When such drugs are withdrawn from a patient receiving Metaglip, the patient should be observed closely for loss of blood glucose control. In vitro binding studies with human serum proteins indicate that glipizide binds differently than tolbutamide and does not interact with salicylate or dicumarol. However, caution must be exercised in extrapolating these findings to the clinical situation and in the use of Metaglip with these drugs.
A potential interaction between oral miconazole and oral hypoglycemic agents leading to severe hypoglycemia has been reported. Whether this interaction also occurs with the intravenous, topical, or vaginal preparations of miconazole is not known. The effect of concomitant administration of fluconazole and glipizide has been demonstrated in a placebo-controlled crossover study in normal volunteers. All subjects received glipizide alone and following treatment with 100 mg of fluconazole as a single oral daily dose for 7 days, the mean percent increase in the glipizide AUC after fluconazole administration was 56.9% (range: 35%-81%).
Metformin Hydrochloride
Furosemide
A single-dose, metformin-furosemide drug interaction study in healthy subjects demonstrated that pharmacokinetic parameters of both compounds were affected by coadministration. Furosemide increased the metformin plasma and blood Cmax by 22% and blood AUC by 15%, without any significant change in metformin renal clearance. When administered with metformin, the Cmax and AUC of furosemide were 31% and 12% smaller, respectively, than when administered alone, and the terminal half-life was decreased by 32%, without any significant change in furosemide renal clearance. No information is available about the interaction of metformin and furosemide when coadministered chronically.
Nifedipine
A single-dose, metformin-nifedipine drug interaction study in normal healthy volunteers demonstrated that coadministration of nifedipine increased plasma metformin Cmax and AUC by 20% and 9%, respectively, and increased the amount excreted in the urine. Tmax and half-life were unaffected. Nifedipine appears to enhance the absorption of metformin. Metformin had minimal effects on nifedipine.
Cationic drugs
Cationic drugs (eg, amiloride, digoxin, morphine, procainamide, quinidine, quinine, ranitidine, triamterene, trimethoprim, or vancomycin) that are eliminated by renal tubular secretion theoretically have the potential for interaction with metformin by competing for common renal tubular transport systems. Such interaction between metformin and oral cimetidine has been observed in normal healthy volunteers in both single- and multiple-dose, metformin-cimetidine drug interaction studies, with a 60% increase in peak metformin plasma and whole blood concentrations and a 40% increase in plasma and whole blood metformin AUC. There was no change in elimination half-life in the single-dose study. Metformin had no effect on cimetidine pharmacokinetics. Although such interactions remain theoretical (except for cimetidine), careful patient monitoring and dose adjustment of Metaglip and/or the interfering drug is recommended in patients who are taking cationic medications that are excreted via the proximal renal tubular secretory system.
Other
In healthy volunteers, the pharmacokinetics of metformin and propranolol and metformin and ibuprofen were not affected when coadministered in single-dose interaction studies.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been conducted with the combined products in Metaglip. The following data are based on findings in studies performed with the individual products.
Glipizide
A 20-month study in rats and an 18-month study in mice at doses up to 75 times the maximum human dose revealed no evidence of drug-related carcinogenicity. Bacterial and in vivo mutagenicity tests were uniformly negative. Studies in rats of both sexes at doses up to 75 times the human dose showed no effects on fertility.
Metformin Hydrochloride
Long-term carcinogenicity studies were performed with metformin alone in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1500 mg/kg/day, respectively. These doses are both approximately 4 times the maximum recommended human daily (MRHD) dose of 2000 mg of the metformin component of Metaglip based on body surface area comparisons. No evidence of carcinogenicity with metformin alone was found in either male or female mice. Similarly, there was no tumorigenic potential observed with metformin alone in male rats. There was, however, an increased incidence of benign stromal uterine polyps in female rats treated with 900 mg/kg/day of metformin alone.
There was no evidence of a mutagenic potential of metformin alone in the following in vitro tests: Ames test (S. typhimurium), gene mutation test (mouse lymphoma cells), or chromosomal aberrations test (human lymphocytes). Results in the in vivo mouse micronucleus test were also negative.
Fertility of male or female rats was unaffected by metformin alone when administered at doses as high as 600 mg/kg/day, which is approximately 3 times the MRHD dose of the metformin component of Metaglip based on body surface area comparisons.
Pregnancy
Teratogenic Effects: Pregnancy Category C
Recent information strongly suggests that abnormal blood glucose levels during pregnancy are associated with a higher incidence of congenital abnormalities. Most experts recommend that insulin be used during pregnancy to maintain blood glucose as close to normal as possible. Because animal reproduction studies are not always predictive of human response, Metaglip should not be used during pregnancy unless clearly needed. (See below.)
There are no adequate and well-controlled studies in pregnant women with Metaglip or its individual components. No animal studies have been conducted with the combined products in Metaglip. The following data are based on findings in studies performed with the individual products.
Glipizide
Glipizide was found to be mildly fetotoxic in rat reproductive studies at all dose levels (5-50 mg/kg). This fetotoxicity has been similarly noted with other sulfonylureas, such as tolbutamide and tolazamide. The effect is perinatal and believed to be directly related to the pharmacologic (hypoglycemic) action of glipizide. In studies in rats and rabbits, no teratogenic effects were found.
Metformin Hydrochloride
Metformin alone was not teratogenic in rats or rabbits at doses up to 600 mg/kg/day. This represents an exposure of about 2 and 6 times the MRHD dose of 2000 mg of the metformin component of Metaglip based on body surface area comparisons for rats and rabbits, respectively. Determination of fetal concentrations demonstrated a partial placental barrier to metformin.
Nonteratogenic Effects
Prolonged severe hypoglycemia (4-10 days) has been reported in neonates born to mothers who were receiving a sulfonylurea drug at the time of delivery. This has been reported more frequently with the use of agents with prolonged half-lives. It is not recommended that Metaglip be used during pregnancy. However, if it is used, Metaglip should be discontinued at least 1 month before the expected delivery date. (See WARNINGS: Pregnancy: Teratogenic Effects: Pregnancy Category C.)
Nursing Mothers
Although it is not known whether glipizide is excreted in human milk, some sulfonylurea drugs are known to be excreted in human milk. Studies in lactating rats show that metformin is excreted into milk and reaches levels comparable to those in plasma. Similar studies have not been conducted in nursing mothers. Because the potential for hypoglycemia in nursing infants may exist, a decision should be made whether to discontinue nursing or to discontinue Metaglip, taking into account the importance of the drug to the mother. If Metaglip is discontinued, and if diet alone is inadequate for controlling blood glucose, insulin therapy should be considered.
Pediatric Use
Safety and effectiveness of Metaglip in pediatric patients have not been established.
Geriatric Use
Of the 345 patients who received Metaglip 2.5 mg/250 mg and 2.5 mg/500 mg in the initial therapy trial, 67 (19.4%) were aged 65 and older while 5 (1.4%) were aged 75 and older. Of the 87 patients who received Metaglip in the second-line therapy trial, 17 (19.5%) were aged 65 and older while 1 (1.1%) was at least aged 75. No overall differences in effectiveness or safety were observed between these patients and younger patients in either the initial therapy trial or the second-line therapy trial, and other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Metformin hydrochloride is known to be substantially excreted by the kidney and because the risk of serious adverse reactions to the drug is greater in patients with impaired renal function, Metaglip should only be used in patients with normal renal function (see CONTRAINDICATIONS, WARNINGS, and CLINICAL PHARMACOLOGY: Pharmacokinetics). Because aging is associated with reduced renal function, Metaglip should be used with caution as age increases. Care should be taken in dose selection and should be based on careful and regular monitoring of renal function. Generally, elderly patients should not be titrated to the maximum dose of Metaglip (see also WARNINGS and DOSAGE AND ADMINISTRATION).
Adverse Reactions
Metaglip
In a double-blind 24-week clinical trial involving Metaglip as initial therapy, a total of 172 patients received Metaglip 2.5 mg/250 mg, 173 received Metaglip 2.5 mg/500 mg, 170 received glipizide, and 177 received metformin. The most common clinical adverse events in these treatment groups are listed in Table 4.
Table 4: Clinical Adverse Events >5% in any Treatment Group, by Primary Term, in Initial Therapy Study
| Number (%) of Patients | ||||
|---|---|---|---|---|
| Adverse Event | Glipizide 5 mg tablets N=170 | Metformin 500 mg tablets N=177 | Metaglip 2.5 mg/250 mg tablets N=172 | Metaglip 2.5 mg/500 mg tablets N=173 |
| Upper respiratory infection | 12 (7.1) | 15 (8.5) | 17 (9.9) | 14 (8.1) |
| Diarrhea | 8 (4.7) | 15 (8.5) | 4 (2.3) | 9 (5.2) |
| Dizziness | 9 (5.3) | 2 (1.1) | 3 (1.7) | 9 (5.2) |
| Hypertension | 17 (10.0) | 10 (5.6) | 5 (2.9) | 6 (3.5) |
| Nausea/vomiting | 6 (3.5) | 9 (5.1) | 1 (0.6) | 3 (1.7) |
In a double-blind 18-week clinical trial involving Metaglip as second-line therapy, a total of 87 patients received Metaglip, 84 received glipizide, and 75 received metformin. The most common clinical adverse events in this clinical trial are listed in Table 5.
Table 5: Clinical Adverse Events >5% in any Treatment Group, by Primary Term, in Second-Line Therapy Study
| Number (%) of Patients | |||
|---|---|---|---|
| Adverse Event | Glipizide 5 mg tabletsa N=84 | Metformin 500 mg tabletsa N=75 | Metaglip 5 mg/500 mg tabletsa N=87 |
| Diarrhea | 11 (13.1) | 13 (17.3) | 16 (18.4) |
| Headache | 5 (6.0) | 4 (5.3) | 11 (12.6) |
| Upper respiratory infection | 11 (13.1) | 8 (10.7) | 9 (10.3) |
| Musculoskeletal pain | 6 (7.1) | 5 (6.7) | 7 (8.0) |
| Nausea/vomiting | 5 (6.0) | 6 (8.0) | 7 (8.0) |
| Abdominal pain | 7 (8.3) | 5 (6.7) | 5 (5.7) |
| UTI | 4 (4.8) | 6 (8.0) | 1 (1.1) |
| a The dose of glipizide was fixed at 30 mg daily; doses of metformin and Metaglip were titrated. | |||
Hypoglycemia
In a controlled initial therapy trial of Metaglip 2.5 mg/250 mg and 2.5 mg/500 mg the numbers of patients with hypoglycemia documented by symptoms (such as dizziness, shakiness, sweating, and hunger) and a fingerstick blood glucose measurement ≤50 mg/dL were 5 (2.9%) for glipizide, 0 (0%) for metformin, 13 (7.6%) for Metaglip 2.5 mg/250 mg, and 16 (9.3%) for Metaglip 2.5 mg/500 mg. Among patients taking either Metaglip 2.5 mg/250 mg or Metaglip 2.5 mg/500 mg, 9 (2.6%) patients discontinued Metaglip due to hypoglycemic symptoms and 1 required medical intervention due to hypoglycemia. In a controlled second-line therapy trial of Metaglip 5 mg/500 mg, the numbers of patients with hypoglycemia documented by symptoms and a fingerstick blood glucose measurement ≤50 mg/dL were 0 (0%) for glipizide, 1 (1.3%) for metformin, and 11 (12.6%) for Metaglip. One (1.1%) patient discontinued Metaglip therapy due to hypoglycemic symptoms and none required medical intervention due to hypoglycemia. (See PRECAUTIONS.)
Gastrointestinal Reactions
Among the most common clinical adverse events in the initial therapy trial were diarrhea and nausea/vomiting; the incidences of these events were lower with both Metaglip dosage strengths than with metformin therapy. There were 4 (1.2%) patients in the initial therapy trial who discontinued Metaglip therapy due to gastrointestinal (GI) adverse events. Gastrointestinal symptoms of diarrhea, nausea/vomiting, and abdominal pain were comparable among Metaglip, glipizide and metformin in the second-line therapy trial. There were 4 (4.6%) patients in the second-line therapy trial who discontinued Metaglip therapy due to GI adverse events.
Overdose
Glipizide
Overdosage of sulfonylureas, including glipizide, can produce hypoglycemia. Mild hypoglycemic symptoms, without loss of consciousness or neurological findings, should be treated aggressively with oral glucose and adjustments in drug dosage and/or meal patterns. Close monitoring should continue until the physician is assured that the patient is out of danger. Severe hypoglycemic reactions with coma, seizure, or other neurological impairment occur infrequently, but constitute medical emergencies requiring immediate hospitalization. If hypoglycemic coma is diagnosed or suspected, the patient should be given a rapid intravenous injection of concentrated (50%) glucose solution. This should be followed by a continuous infusion of a more dilute (10%) glucose solution at a rate that will maintain the blood glucose at a level above 100 mg/dL. Patients should be closely monitored for a minimum of 24 to 48 hours, since hypoglycemia may recur after apparent clinical recovery. Clearance of glipizide from plasma would be prolonged in persons with liver disease. Because of the extensive protein binding of glipizide, dialysis is unlikely to be of benefit.
Metformin Hydrochloride
Overdose of metformin hydrochloride has occurred, including ingestion of amounts >50 g. Hypoglycemia was reported in approximately 10% of cases, but no causal association with metformin hydrochloride has been established. Lactic acidosis has been reported in approximately 32% of metformin overdose cases (see WARNINGS). Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful for removal of accumulated drug from patients in whom metformin overdosage is suspected.
Dosage and Administration
General Considerations
Dosage of Metaglip must be individualized on the basis of both effectiveness and tolerance while not exceeding the maximum recommended daily dose of 20 mg glipizide/2000 mg metformin. Metaglip should be given with meals and should be initiated at a low dose, with gradual dose escalation as described below, in order to avoid hypoglycemia (largely due to glipizide), reduce GI side effects (largely due to metformin), and permit determination of the minimum effective dose for adequate control of blood glucose for the individual patient.
With initial treatment and during dose titration, appropriate blood glucose monitoring should be used to determine the therapeutic response to Metaglip and to identify the minimum effective dose for the patient. Thereafter, HbA1c should be measured at intervals of approximately 3 months to assess the effectiveness of therapy. The therapeutic goal in all patients with type 2 diabetes is to decrease FPG, PPG, and HbA1c to normal or as near normal as possible. Ideally, the response to therapy should be evaluated using HbA1c, which is a better indicator of long-term glycemic control than FPG alone.
No studies have been performed specifically examining the safety and efficacy of switching to Metaglip therapy in patients taking concomitant glipizide (or other sulfonylurea) plus metformin. Changes in glycemic control may occur in such patients, with either hyperglycemia or hypoglycemia possible. Any change in therapy of type 2 diabetes should be undertaken with care and appropriate monitoring.
Metaglip in Patients with Inadequate Glycemic Control on Diet and Exercise Alone
For patients with type 2 diabetes whose hyperglycemia cannot be satisfactorily managed with diet and exercise alone, the recommended starting dose of Metaglip is 2.5 mg/250 mg once a day with a meal. For patients whose FPG is 280 mg/dL to 320 mg/dL a starting dose of Metaglip 2.5 mg/500 mg twice daily should be considered. The efficacy of Metaglip in patients whose FPG exceeds 320 mg/dL has not been established. Dosage increases to achieve adequate glycemic control should be made in increments of 1 tablet per day every 2 weeks up to maximum of 10 mg/1000 mg or 10 mg/2000 mg Metaglip per day given in divided doses. In clinical trials of Metaglip as initial therapy, there was no experience with total daily doses >10 mg/2000 mg per day.
Metaglip in Patients with Inadequate Glycemic Control on a Sulfonylurea and/or Metformin
For patients not adequately controlled on either glipizide (or another sulfonylurea) or metformin alone, the recommended starting dose of Metaglip is 2.5 mg/500 mg or 5 mg/500 mg twice daily with the morning and evening meals. In order to avoid hypoglycemia, the starting dose of Metaglip should not exceed the daily doses of glipizide or metformin already being taken. The daily dose should be titrated in increments of no more than 5 mg/500 mg up to the minimum effective dose to achieve adequate control of blood glucose or to a maximum dose of 20 mg/2000 mg per day.
Patients previously treated with combination therapy of glipizide (or another sulfonylurea) plus metformin may be switched to Metaglip 2.5 mg/500 mg or 5 mg/500 mg; the starting dose should not exceed the daily dose of glipizide (or equivalent dose of another sulfonylurea) and metformin already being taken. The decision to switch to the nearest equivalent dose or to titrate should be based on clinical judgment. Patients should be monitored closely for signs and symptoms of hypoglycemia following such a switch and the dose of Metaglip should be titrated as described above to achieve adequate control of blood glucose.
Specific Patient Populations
Metaglip is not recommended for use during pregnancy or for use in pediatric patients. The initial and maintenance dosing of Metaglip should be conservative in patients with advanced age, due to the potential for decreased renal function in this population. Any dosage adjustment requires a careful assessment of renal function. Generally, elderly, debilitated, and malnourished patients should not be titrated to the maximum dose of Metaglip to avoid the risk of hypoglycemia. Monitoring of renal function is necessary to aid in prevention of metformin-associated lactic acidosis, particularly in the elderly. (See WARNINGS.)
How Supplied
Metaglip™(glipizide and metformin HCl) Tablets
Metaglip 2.5 mg/250 mg tablet is a pink oval-shaped, biconvex film-coated tablet with "BMS" debossed on one side and "6081" debossed on the opposite side.
Metaglip 2.5 mg/500 mg tablet is a white oval-shaped, biconvex film-coated tablet with "BMS" debossed on one side and "6077" debossed on the opposite side.
Metaglip 5 mg/500 mg tablet is a pink oval-shaped, biconvex film-coated tablet with "BMS" debossed on one side and "6078" debossed on the opposite side.
| Metaglip | NDC 0087-xxxx-xx for unit of use | |
|---|---|---|
| Glipizide (mg) | Metformin hydrochloride (mg) | Bottle of 100 |
| 2.5 | 250 | 6081-31 |
| 2.5 | 500 | 6077-31 |
| 5.0 | 500 | 6078-31 |
Storage
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature.]
Metaglip™ is a trademark of Merck Santé S.A.S., an associate of Merck KGaA of Darmstadt, Germany. Licensed to Bristol-Myers Squibb Company.
GLUCOPHAGE® is a registered trademark of Merck Santé S.A.S., an associate of Merck KGaA of Darmstadt, Germany. Licensed to Bristol-Myers Squibb Company.
GLUCOTROL® is a registered trademark of Pfizer Inc.
Distributed by:
Bristol-Myers Squibb Company
Princeton, NJ 08543 USA
Last Updated: 07/09
Metaglip, glipizide and metformin hydrochloride, patient information sheet (in plain English)
Detailed Info on Signs, Symptoms, Causes, Treatments of Diabetes
The information in this monograph is not intended to cover all possible uses, directions, precautions, drug interactions or adverse effects. This information is generalized and is not intended as specific medical advice. If you have questions about the medicines you are taking or would like more information, check with your doctor, pharmacist, or nurse.
back to: Browse all Medications for Diabetes
APA Reference
Staff, H.
(2009, July 31). Metaglip Treatment for Diabetes - Metaglip Full Prescribing Information, HealthyPlace. Retrieved
on 2026, March 30 from https://www.healthyplace.com/diabetes/medications/metaglip-glipizide-and-metformin-hcl-diabetic-treatment