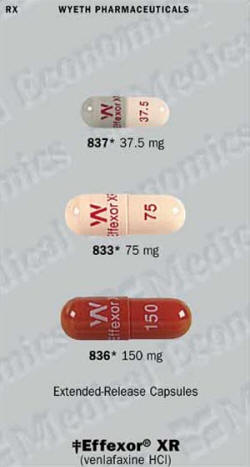
Effexor XR (Venlafaxine) Patient Information
Find out why Effexor XR is prescribed, Effexor XR side effects, Effexor XR warnings, effects of Effexor XR during pregnancy, more - in plain English.
Generic name: Venlafaxine hydrochloride
Other brand name: Effexor XR
Pronounced: ef-ECKS-or
Effexor (venlafaxin) XR Full Prescribing Information
Why is Effexor XR prescribed?
Effexor is prescribed for the treatment of depression--that is, a continuing depression that interferes with daily functioning. The symptoms usually include changes in appetite, sleep habits, and mind/body coordination, decreased sex drive, increased fatigue, feelings of guilt or worthlessness, difficulty concentrating, slowed thinking, and suicidal thoughts.
Effexor XR is also prescribed to relieve abnormal anxiety (generalized anxiety disorder). This problem is marked by persistent anxiety for a period of at least 6 months, accompanied by at least 3 of these 6 symptoms: restlessness, fatigue, poor concentration, irritability, muscle tension, and sleep disturbances.
Effexor must be taken 2 or 3 times daily. The extended-release form, Effexor XR, permits once-a-day dosing.
Most important fact about Effexor XR
Serious, sometimes fatal reactions have occurred when Effexor is used in combination with other drugs known as MAO inhibitors, including the antidepressants Nardil and Parnate. Never take Effexor with one of these drugs; and do not begin therapy with Effexor within 14 days of discontinuing treatment with one of them. Also, allow at least 7 days between the last dose of Effexor and the first dose of an MAO inhibitor.
How should you take Effexor XR?
Take Effexor with food, exactly as prescribed. It may take several weeks before you begin to feel better. Your doctor should check your progress periodically.
Take Effexor XR once at the same time each day. Swallow the capsule whole with water. Do not divide, crush, or chew it.
--If you miss a dose...
It is not necessary to make it up. Skip the missed dose and continue with your next scheduled dose. Do not take 2 doses at once.
--Storage instructions...
Store in a tightly closed container at room temperature. Protect from excessive heat and moisture.
What side effects may occur with Effexor XR?
Side effects cannot be anticipated. If any develop or change in intensity, tell your doctor as soon as possible. Only your doctor can determine if it is safe for you to continue taking Effexor.
-
More common Effexor XR side effects may include: Abnormal dreams, abnormal ejaculation or orgasm, anxiety, appetite loss, blurred vision, chills, constipation, diarrhea, dizziness, dry mouth, frequent urination, flushing, gas, headache, impotence, infection, insomnia, muscle tension, nausea, nervousness, rash, sleepiness, sweating, tingling feeling, tremor, upset stomach, vomiting, weakness, yawning
-
Less common side effects may include: Abnormal taste, abnormal thinking, agitation, chest pain, confusion, decreased sex drive, depression, dilated pupils, dizziness upon standing up, high blood pressure, itching, loss of identity, rapid heartbeat, ringing in the ears, trauma, twitching, urinary problems, weight loss
A wide variety of very rare symptoms possibly related to Effexor have also been reported. If you develop any new or unusual problems, let your doctor know about it.
Why should Effexor XR not be prescribed?
Never take Effexor while taking other drugs known as MAO inhibitors. (See "Most important fact about this drug.") Also avoid this drug if it has ever given you an allergic reaction.
Special warnings about Effexor XR
Your doctor will prescribe Effexor with caution if you have high blood pressure, heart, liver, or kidney disease or a history of seizures or mania (extreme agitation or excitability). You should discuss all of your medical problems with your doctor before taking Effexor.
Effexor sometimes causes an increase in blood pressure. If this happens, your doctor may need to reduce your dose or discontinue the drug.
Effexor also tends to increase the heart rate, especially at higher doses. Use Effexor with caution if you've recently had a heart attack, suffer from heart failure, or have an overactive thyroid gland.
Antidepressants such as Effexor may cause fluid retention, especially if you are an older adult.
Effexor may cause you to feel drowsy or less alert and may affect your judgment. Therefore, avoid driving or operating dangerous machinery or participating in any hazardous activity that requires full mental alertness until you know how this drug affects you.
Your doctor will check you regularly if you have glaucoma (high pressure in the eye), or you are at risk of developing it.
If you have ever been addicted to drugs, tell your doctor before you start taking Effexor.
If you develop a skin rash or hives while taking Effexor, notify your doctor. Effexor may also cause bleeding or bruising of the skin.
Do not stop taking the drug without consulting your doctor. If you stop suddenly, you may have withdrawal symptoms, even though this drug does not seem to be habit-forming. Your doctor will have you taper off gradually.
The safety and effectiveness of Effexor have not been established in children under 18 years of age.
Possible food and drug interactions when taking Effexor XR
Combining Effexor with MAO inhibitors could cause a fatal reaction. (See "Most important fact about this drug.")
Although Effexor does not interact with alcohol, the manufacturer recommends avoiding alcohol while taking this medication.
If you have high blood pressure or liver disease, or are elderly, check with your doctor before combining Effexor with cimetidine (Tagamet).
 Effexor does not interact with Lithium or Valium. However, you should consult your doctor before combining Effexor with other drugs that affect the central nervous system, including narcotic painkillers, sleep aids, tranquilizers, antipsychotic medicines such as Haldol, and other antidepressants such as Tofranil.
Effexor does not interact with Lithium or Valium. However, you should consult your doctor before combining Effexor with other drugs that affect the central nervous system, including narcotic painkillers, sleep aids, tranquilizers, antipsychotic medicines such as Haldol, and other antidepressants such as Tofranil.
Effexor has been found to reduce blood levels of the HIV drug Crixivan. It's best to check with your doctor before combining Effexor with any other drug or herbal product.
Special information if you are pregnant or breastfeeding
The effects of Effexor during pregnancy have not been adequately studied. If you are pregnant or are planning to become pregnant, tell your doctor immediately. Effexor should be used during pregnancy only if clearly needed.
If Effexor is taken shortly before delivery, the baby may suffer withdrawal symptoms. It's also known that Effexor appears in breast milk and could cause serious side effects in a nursing infant. You'll need to choose between nursing your baby or continuing your treatment with Effexor.
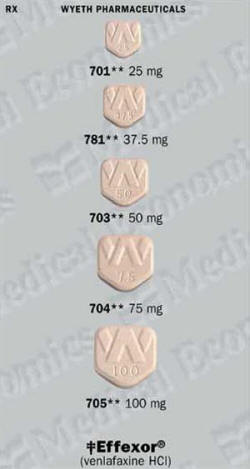
Recommended dosage of Effexor XR
EFFEXOR
The usual starting dose is 75 milligrams a day, divided into 2 or 3 smaller doses, and taken with food. If needed, your doctor may gradually increase your daily dose in steps of no more than 75 milligrams at a time up to a maximum of 375 milligrams per day.
If you have kidney or liver disease or are taking other medications, your doctor will adjust your dosage accordingly.
EFFEXOR XR
For both depression and anxiety the usual starting dose is 75 milligrams once daily, although some people begin with a dose of 37.5 milligrams for the first 4 to 7 days. Your doctor may gradually increase the dose, in steps of no more than 75 milligrams, up to a maximum of 225 milligrams daily. As with regular Effexor, the doctor will make adjustments in your dosage if you have kidney or liver disease.
Overdosage of Effexor XR
An overdose of Effexor, combined with other drugs or alcohol, can be fatal. If you suspect an overdose, seek medical attention immediately.
- Symptoms of Effexor overdose include: Sleepiness, vertigo, rapid or slow heartbeat, low blood pressure, seizures, coma
Effexor (venlafaxin) XR Full Prescribing Information
Detailed Info on Signs, Symptoms, Causes, Treatments of Depression
Detailed Info on Signs, Symptoms, Causes, Treatments of Anxiety Disorders
APA Reference
Staff, H.
(2009, January 4). Effexor XR (Venlafaxine) Patient Information, HealthyPlace. Retrieved
on 2024, April 26 from https://www.healthyplace.com/other-info/psychiatric-medications/effexor-xr-venlafaxine-patient-information